Sixty percent of US life sciences CEOs expect to actively pursue M&A over the next 12 months, according to the latest EY-Parthenon CEO Outlook Survey, though many are skeptical that there will be an uptick in megadeals.
While many life sciences companies, such as large pharmaceutical and diagnostic companies, have a methodology and ways of working through bolt-on deals, our experience shows that challenges exist throughout the deal lifecycle.
The following mix of strategic and tactical actions can help life sciences companies deliver the outcomes they need from their bolt-on acquisitions, including expected return on investment capital:
1. Deal rationale and integration strategy
Obtain strong alignment from the board down to the working teams regarding why you are buying the asset or business. Things to consider:
- Is it an adjacent TA?
- Does the management team have critical knowledge or technology you are bolting-on to your current operating model?
Once aligned on the deal rationale, communicate it early and often to all stakeholder groups before moving to the integration strategy
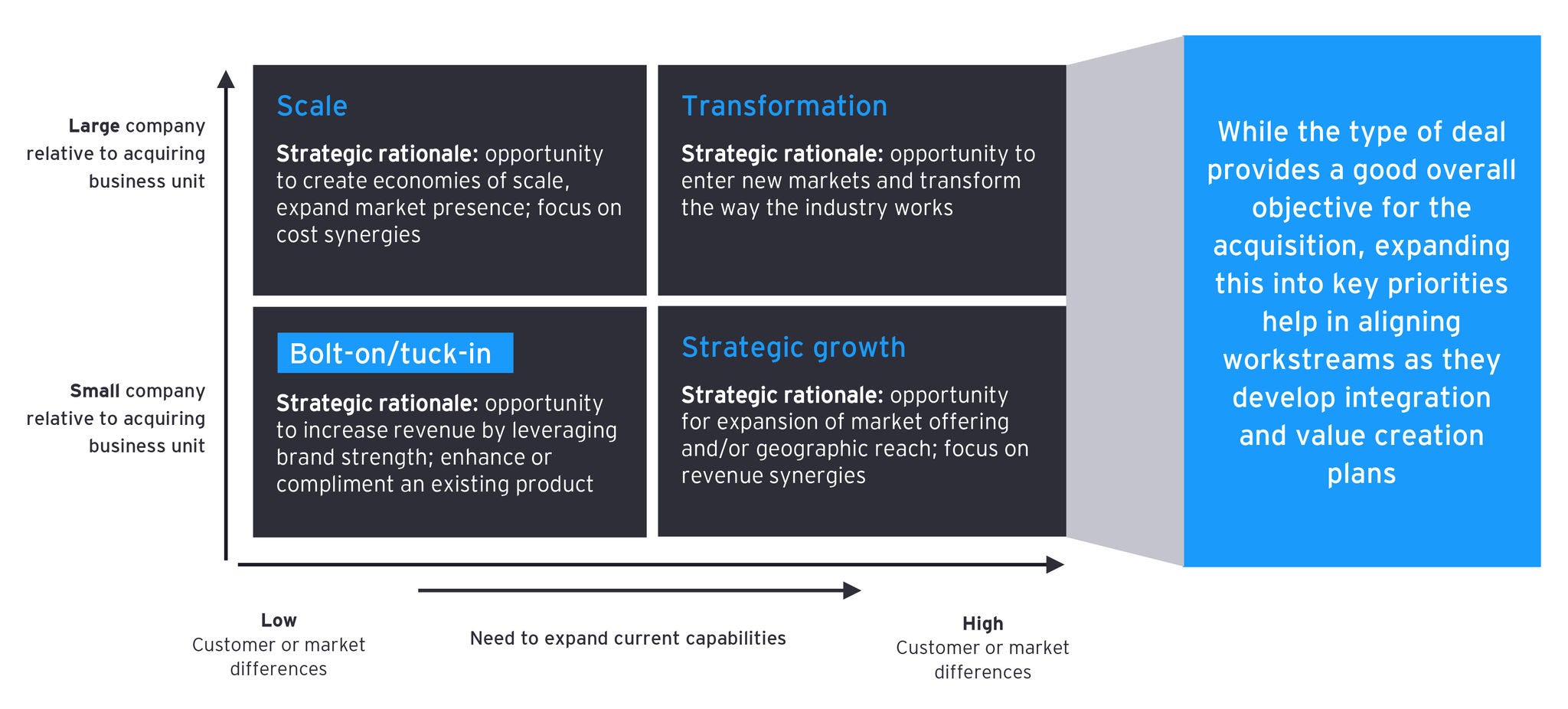
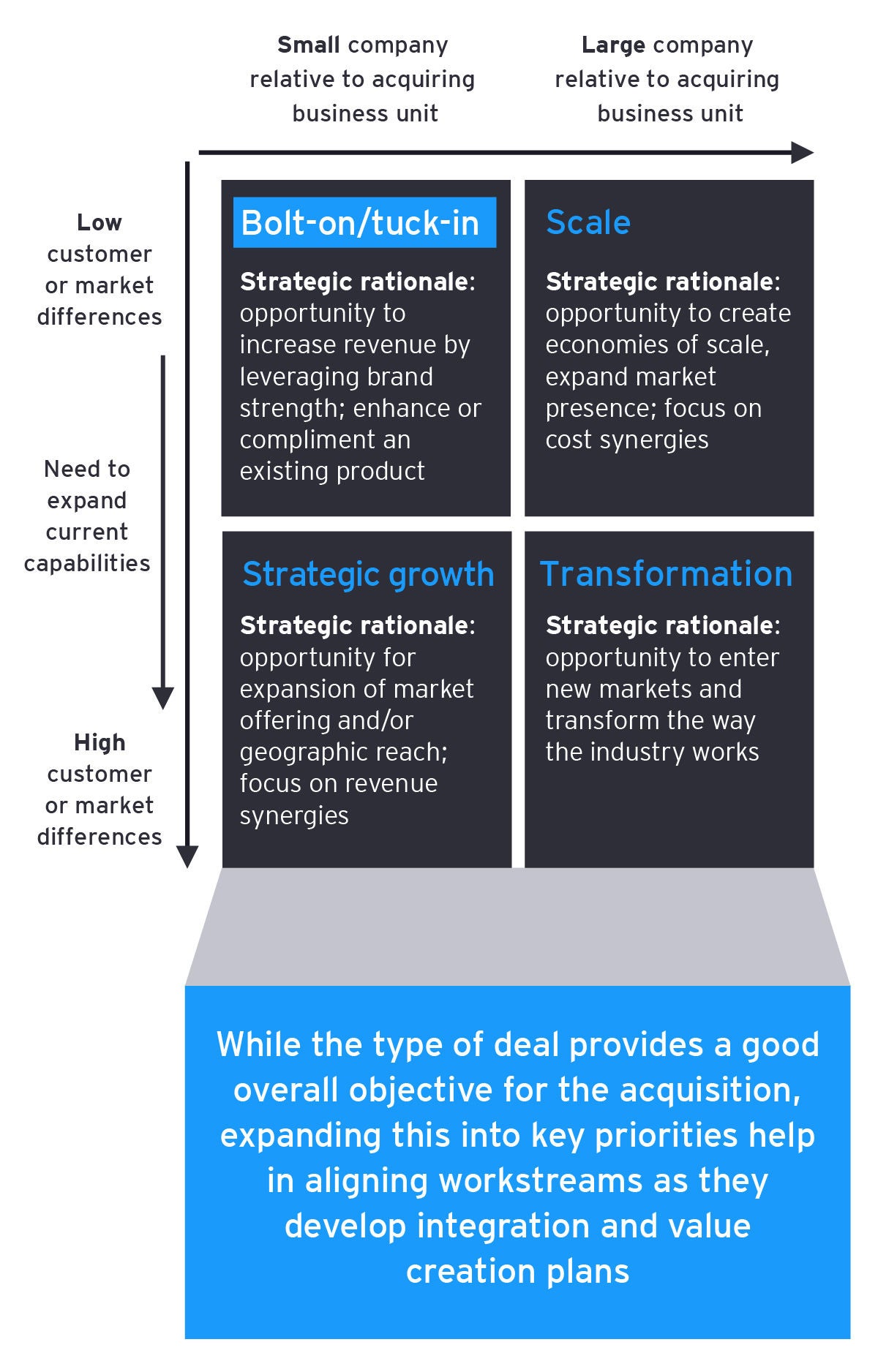
In regard to the deal rationale (or “investment thesis”), the first key step is to define the deal type within your deal archetypes. Some companies only have two: bolt-on or strategic (sometimes referred to as “transformational”). EY-Parthenon teams think about deals through the four deal types below. Based on rationale and deal type, memorialize and communicate the integration strategy.